Yeast: a single-celled
microorganism
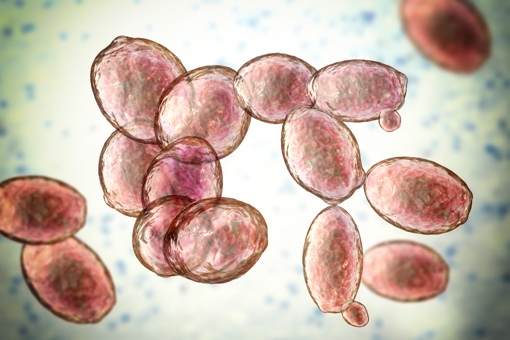
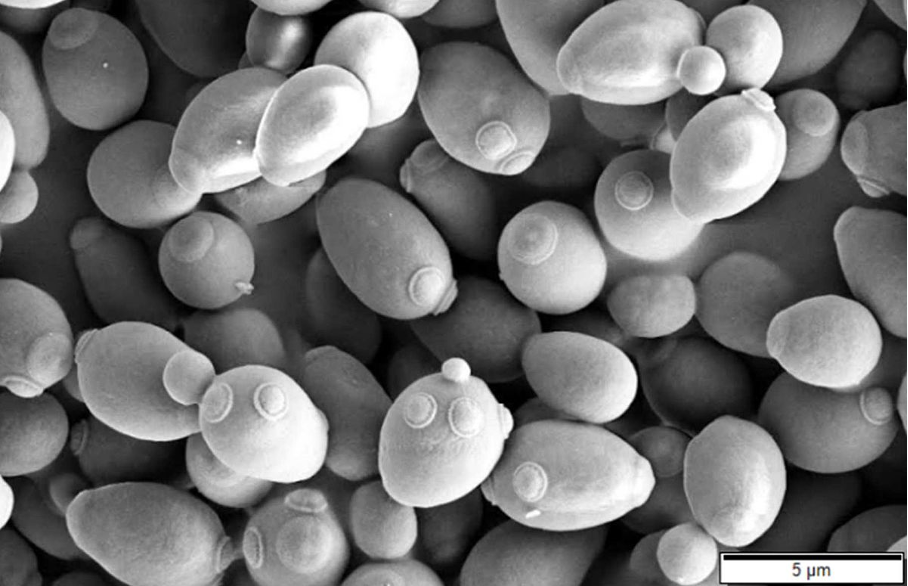
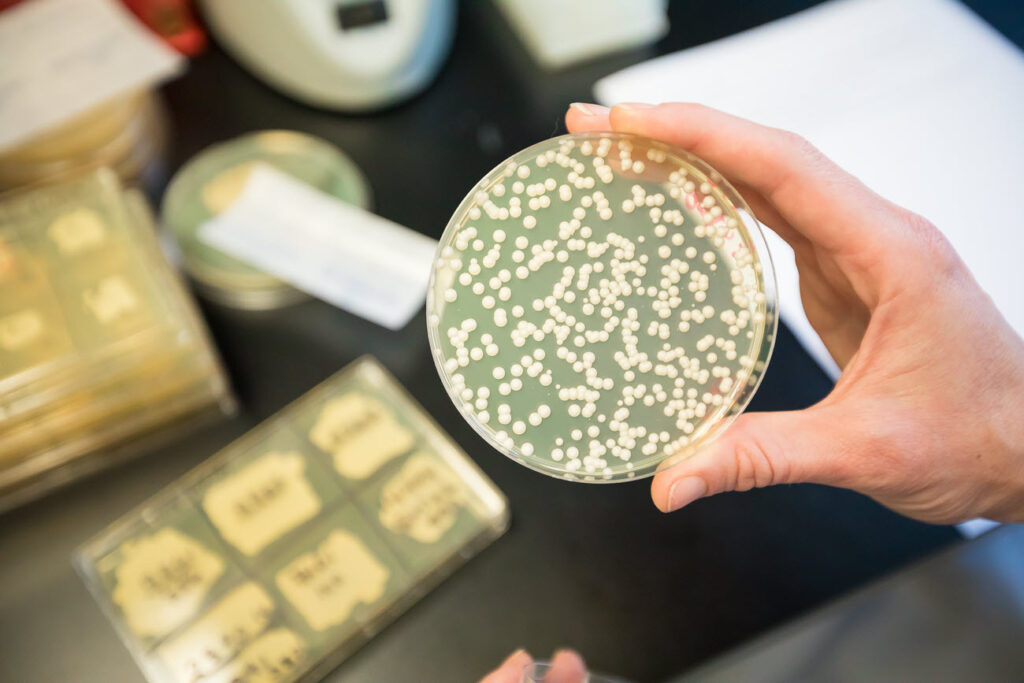
Did you know that one cubic centimetre of fresh yeast, or about one gram, can contain up to 10 billion living yeast cells!
These cells are microscopic fungi: unlike other microorganisms such as bacteria or viruses, yeasts are eukaryotic cells.
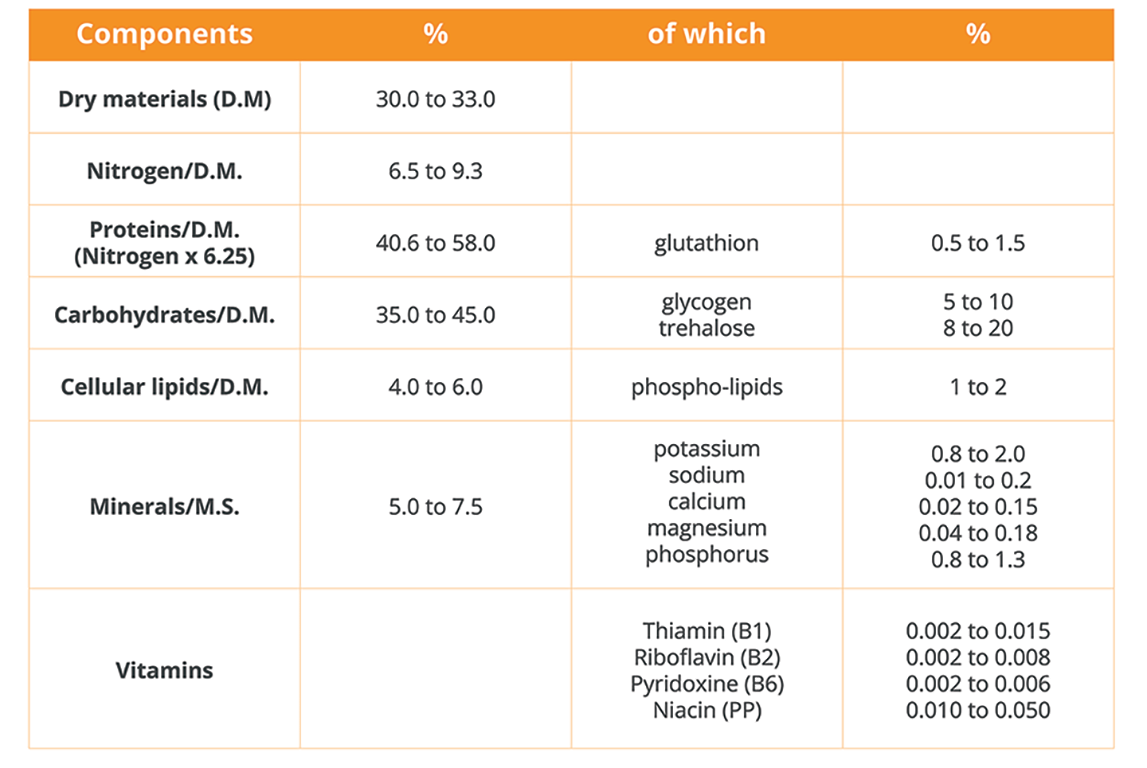
This means that they have a clearly defined nucleus and the genetic material necessary for the biosynthesis of complex molecules such as enzymes, beta-glucans, mannoproteins, and lipids.
Thanks to these precious molecules, yeast is able to ferment and transform matter into other useful substances.